FAO Asks Countries to Simplify Regulation for Cultivated Meat & Precision Fermentation
The UN Food and Agriculture Organization is calling on governments to streamline their regulatory frameworks for cultivated proteins and precision-fermented foods.
As novel foods like cultivated meat and precision-fermented ingredients make progress on their path to mainstream adoption, the UN Food and Agriculture Organization (FAO) is weighing in on their regulation.
In a new legal paper, the food body draws on frameworks like the Codex Alimentarius (established by the FAO and the World Health Organization) and World Trade Organization agreements to highlight the gaps and challenges in regulating these novel foods, arguing that this isn’t just a technical issue, it’s “also a political and a legal one”.
Legislation can provide countries with the ability to control the market entry of these products, as well as set the framework to implement conscious policy decisions over their development. However, there exists very little guidance on relevant legislation in this area.
Most of the focus, the FAO argues, is on terminology and labelling, with the EU being the most prominent case. There’s limited exploration of food safety and pre-market approval processes.
“Notably absent from the current literature is a substantial analysis of how cell-based food products integrate into general systems of food law and food safety regulation. There is little or no research on the impact of product-specific regulatory frameworks or necessary adaptations for food operators,” the report states.
FAO addresses novel food tastings, bans and labelling

The legal paper analyses regulatory approaches in regional and national legislation and outlines that existing frameworks for food safety – particularly those addressing new food sources and production systems – would generally apply to cell-cultivated and precision-fermented products too.
It notes that legislation should clearly define which authorities will regulate, monitor and control the safety of these novel foods. This includes the development and oversight of food, safety standards, quality assurance, and the approval process for market entry.
The US is a good example here: the Food and Drug Administration oversees the pre-harvest stage of production (like cell collection, banking and proliferation) across all cultivated protein species, before transferring responsibility to the Department of Agriculture for the post-harvest phase (like processing, packaging and labelling) for most categories, barring seafood.
Further, the FAO suggests that pre-approval tastings for these foods deserve special attention as a legal issue, since they serve as a “provisional bridge between experimental production and market entry”, but aren’t currently accounted for in most pre-market authorisation frameworks.
The paper acknowledges how some countries (like Italy) have banned or considered restrictions on cultivated meat, which have justified these efforts as a precautionary measure in the absence of comprehensive risk assessments or awaiting regulatory precedents from other nations.
“However, these country decisions and grounds must align with the country’s international and/or regional legal obligations,” says the UN body.
It argues that transparent and accurate labelling is key for these products to prevent consumer confusion or misinformation, although surveys have consistently shown that labelling meat alternatives as ‘meat’ doesn’t mislead the public.
“Key issues around labelling include the terminology used to describe the product to make clear that it is produced through these methods, the naming of the species that was used, conveying increasingly complex messages about allergens, ingredients used in production and the involvement of GMOs,” says the FAO.
How countries can advance regulatory frameworks for future foods
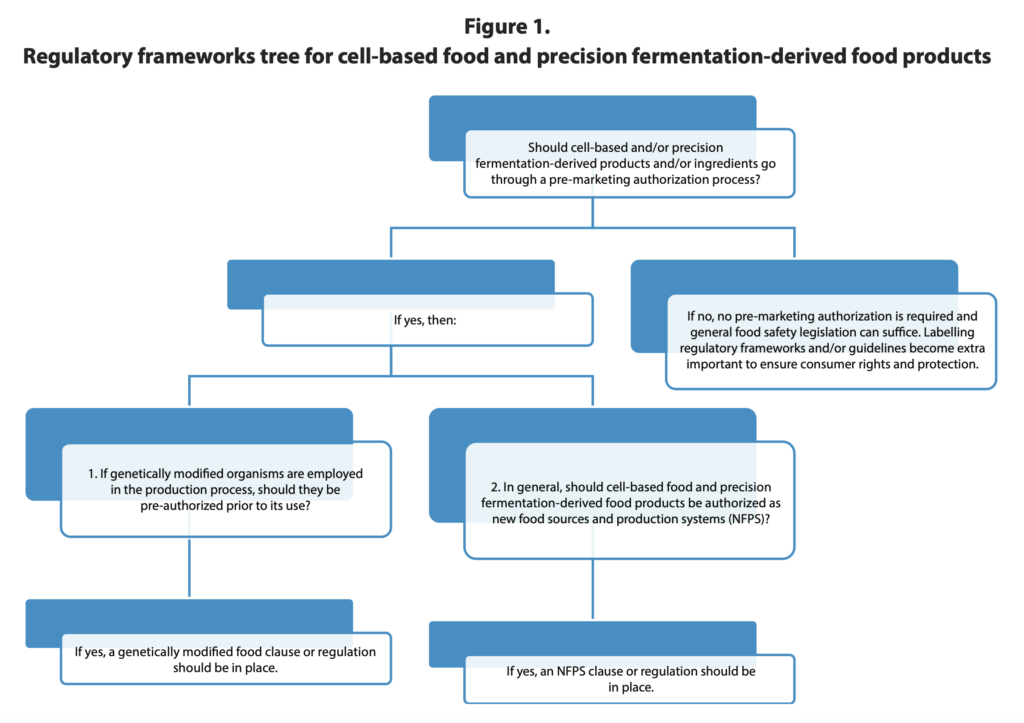
Looking ahead, the FAO outlines several priority actions for countries that want to solidify their regulatory frameworks for cultivated and precision-fermented proteins, fats, and the like.
Authorities should assess and clarify how existing regulatory frameworks apply to novel foods. This may be necessary for cases where current regulations have some ambiguity over their suitability for these products, helping provide legal certainty for food manufacturers and ensuring that food safety standards for conventional products are appropriately adjusted.
The FAO has urged governments to consider whether additional pre-market approval frameworks for cultivated and precision-fermented foods are required beyond general food safety regulations. It suggests that countries have various options and modalities to consider if they do want to implement supplementary processes.
In some cases, specific regulatory elements may need to be developed or updated, such as those relating to food labelling and nomenclature, or lists of permissible additives, enzymes and processing aids that reflect the needs of these new technologies.
“In some cases, in addition to other options, authorities may want to develop clear supporting guidelines for the production, labelling, and market authorisation of these products, as well as producing good manufacturing practices, good cell culture practices, good hygiene practices to help guide producers and set standards,” says the FAO.
Further, the organisation is advocating for international bodies like the Codex Alimentarius to continue discussions on clear definitions and guidelines for these products, which will facilitate smoother international trade and reduce legal uncertainties for food producers. The harmonisation of these new foods is thus an important pathway to consider.
Finally, the FAO states that regulatory frameworks must be developed through broad consultations to reflect all relevant perspectives. This means not just producers, consumers, industry, civil society groups, and governments, but also organisations focused on the environment and animal rights, and agricultural workers and unions.
“Input from biotechnology experts, public health authorities, and trade associations will ensure the inclusion of innovation, health assessments, and smooth market integration,” the paper reads. “This approach ensures that regulations align with societal values and address key concerns around safety, transparency, ethics, and sustainability.”



